Big enough to deliver and small enough to care
HealthTech developers, including drug developers and medical device manufacturers are facing many challenges in clinical research:
- Lack of patient engagement in study design
- Unfamiliar with the new EU Clinical Trials Regulations (EU CTR) and its submission portal (CTIS)
- Difficult to have a customizable clinical study tool
- Challenging to manage all stakeholders in clinical studies
- Unfamiliar with early access opportunities
We support the development of drugs and medical devices for unmet medical needs to overcome these challenges and improve the health and quality of life of patients.
End-to-end clinical research services
At VCLS, we focus on designing, implementing, managing and reporting clinical studies with proprietary digital tools for the HealthTech industry.
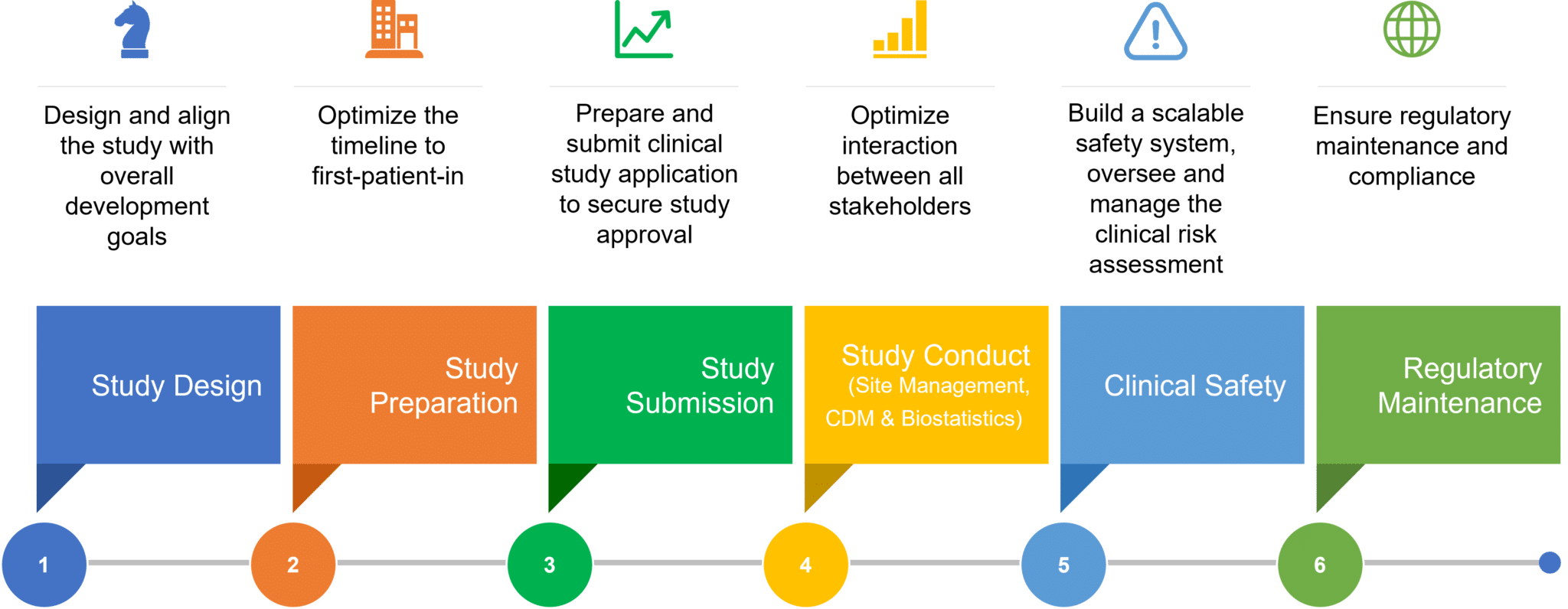
Patient-centric protocol design
- Align the study with overall medical, access to market & regulatory goals
- Author patient-centric protocol and liaise with patient organizations for optimal patient engagement
Study submission & operation
- Prepare and submit clinical trial applications on the new EU CTR and portal to secure timely study approval
- Study conduct, data management & analysis
- Clinical safety
Proprietary clinical study tools & project management team
- Proprietary CTMS, including ePRO, eCRF that are customizable per clients’ needs
- Robust QMS
- Agile project management team with a blend of scientific, clinical and regulatory backgrounds
Strong expertise in oncology and medical devices
- Hands-on therapeutic expertise, product knowledge and knowhow in oncology and medical devices
- A science-savvy interdisciplinary experience

I really enjoy working with the VCLS team because I know that they always have things under control. They are prompt to escalate any issues and implement team decisions quickly. They always offer their expertise and collaborate well with all parties (sites, CRAs, sponsor, etc) to find mutually agreeable resolutions to keep projects moving forward. And they’re all really nice and funny, so every meeting is engaging.
Director of Clinical Operations, US-based emerging biotech focusing on rare disease



